Debunking the Climate Scam
Billions of Dollars -
Greedy Green Corporations -
No Warming For Two decades -
Bought and Paid For Organizations

5000 Years Of Climate Change From Greenland Ice Cores

November 3, 2009
Gordon J. Fulks, PhD
(Preliminary Draft)
Disclaimer: This paper was prepared without compensation. The author is not
employed by any of the parties having a financial interest in this topic.
Gordon J. Fulks and Associates
La Center, Washington USA
SUMMARY
Despite a sophisticated effort to promote a new scare about “ocean acidification” that is basically an
offshoot of the Global Warming scare, there is no scientific evidence that the new scare has any
validity. Our oceans are mildly alkaline or basic with an average pH of about 8 that can vary in time
and/or location by about 0.2 up or down. That remarkable stability is maintained by the buffering
action of calcium carbonate which is widely abundant. Increasing atmospheric carbon dioxide from
whatever source (man-
the atmospheric carbon dioxide dissolves in sea water. But the strong buffering action of calcium
carbonate will keep the average pH close to 8.0. Hence no significant change in ocean alkalinity is
expected. The oceans have not and will not become acidic over large areas as claimed by Al Gore.
The only concern might be the health of some ocean creatures that use calcium carbonate (especially
the aragonite crystalline form) for their shells. If there is insufficient calcium otherwise available in a
particular area to buffer the additional CO2 coming into solution, then the sea creatures in that area
could lose some shell material to the buffering process. This will not threaten the overall survival of
corals because we know they have survived far greater levels of atmospheric carbon dioxide than man
can ever create by burning fossil fuels. Corals are very survivable creatures as evidenced by their
existence on earth for hundreds of millions of years, far longer than man.
IMPLICATIONS FOR POLICY MAKERS
An essential conclusion of this paper for policy makers is that they should not reward applicants for
grants who use scare tactics in their pursuit of research funds. Doing so only encourages others to use
the same approach. Also, it is unlikely that those who exaggerate the value of their research as
“absolutely vital” to the survival of the planet will do an honest job with their research.
Ocean research should continue at appropriate levels (not expanded levels), but only with those who do
not employ scare tactics. The peer review process needs to be strengthened such that papers for
publication are reviewed by competitors of those seeking to publish, not just those who are friends or
close associates. Journals (especially Science and Nature) need to enforce rules about data archiving
and release, so critics can see if authors are really following accepted standards of academic excellence.
Those caught cheating need to punished. If Science and/or Nature continue favoring one line of
reasoning (eg., Global Warming) over all others, their access to federal money should be restricted or
Terminated.
Climate science needs to be returned to the fundamental dialectic of all science. Theory and
observation must be clearly separate pursuits that compliment each other and enforce objectivity.
Allowing theory and simulations to become the new standard of rigor is to permit the disintegration of
objectivity. Allowing government bureaucrats to decide which theories and simulations are worthy of
funding further erodes objectivity. The ultimate arbiter in all questions of science is real evidence
obtained by truly independent researchers. The standard of objectivity in all human pursuits is
independence from conflicts of interest, especially financial conflicts of interest.
BACKGROUND
Over the last twenty plus years, we have seen a proliferation of scientific scams based on Al Gore's
theory of Anthropogenic Global Warming caused by man-
characteristics, including a thread of truth woven into a narrative about an apocalypse that sounds both
convincing and frightening to those without scientific training.
To many of those with scientific training it has become a dream come true with almost unlimited
government funding available. All these scientists need do is profess a belief in Global Warming and
convince a government agency that their studies address “the problem.” Of course their studies never
address crucial questions such as the role man is actually playing in the changes they observe. Even if
they observe changes that clearly indicate cooling rather than warming despite an increase in
atmospheric carbon dioxide, they go on as though everything is fine with the underlying hypothesis.
Amongst those who owe their jobs to AGW, there is no doubt that Al Gore is correct.
To focus attention on their area of expertise and build public support for their efforts, too many
scientists have taken to issuing press releases that highlight their work and point to some “grave
concern” about a rapidly approaching “tipping point.” Of course, we are not quite there yet and just
might be able to avoid the apocalypse if the government significantly increases their funding. The
grave concern could be anything from the melting of the Arctic Tundra, the melting of the Arctic Pack
Ice, the melting of the Greenland Ice Cap, warming in West Antarctica, and rising sea levels to a
particular species migrating or under stress and the oceans acidifying to the point where much of the
life within will be destroyed. The point they wish to drive home is that we just might be able to save
the planet if we support their research efforts. The problem is never so dire that all hope is lost or so
inconsequential that it can be ignored. Just support more money for them, and the planet will be saved!
Another advantage to having a long list of concerns is purely practical. When the climate stubbornly
refuses to warm as the UN IPCC maintains it must, Alarmists can easily shift attention to something
else until the climate naturally cycles back to a warm phase.
Just a little knowledge of the natural phenomena under consideration is necessary to spot the essential
hoax: on a fluid planet with vast oceans and atmosphere, many physical and biological processes are
constantly changing for completely natural reasons. The only thing that would be truly unnatural
would be for everything to remain constant. Yet those without scientific training are easily sold on the
idea that change is unnatural and must be avoided at all costs. That's rubbish and dangerous.
When finally cornered, many proponents of the apocalypse fall back on the “zero risk” argument. They
argue that we should follow their recommendations for reducing per capita carbon emissions to levels
not seen since the 19th century to eliminate ALL RISK. That betrays a lack of understanding of risk,
which is never zero. If we were to return to a primitive 19th century lifestyle without the energy that
drives our modern economy today, our life expectancy could drop sharply to what is was then. The
world as a whole would be far worse off, because a modern mechanized society is necessary to support
the world's present population. A return to the past, even if it were possible, is not progress, it is
disaster.
THE ACID OCEAN HYPOTHESIS according to Gore
Here is the hypothesis from Al Gore:
“Carbon dioxide pollution is changing the very chemistry of our oceans. Ocean acidification is already
underway and is accelerating. A recent paper published in the journal Science described how the
seawater off the coast of Northern California has become so acidic from CO2 that it is now corrosive.
To give some sense of perspective, for the last 44 million years, the average pH of the water has been
8.2. The scientists at Scripps measured levels off the north coast of California and Oregon at a pH of
7.75. Coral polyps that make reefs and everything that makes a shell are now beginning to suffer from
a kind of osteoporosis because of the 25 million tons of CO2 absorbed by the oceans every 24 hours.”
This is mostly nonsense. First of all, the oceans are mildly basic or alkaline not acidic, with an average
pH of about 8. Figure 1 shows the pH scale with the pH of common solutions indicated. Freshly
distilled water is neutral at 7. Rain water is much more acidic at 5.6 because of dissolved CO2, beer
still more acidic at 4.3, and carbonated beverages still more acidic because they contain phosphoric
acid in addition to their carbonation. Soda can be nearly as acidic as lemon juice.
The oceans are almost as basic as a solution of baking soda (sodium bicarbonate) but far from as
corrosive as bleach or lye. If the ocean pH is shifting at all, it is shifting ever so sightly toward neutral.
There is nothing in the scientific literature to suggest in any way under any circumstances that the
oceans will become acidic. That's just Gore's imagination running wild. Scientists do talk about
decreases in pH moving the solution toward acidic or increases moving it toward basic. In other words,
diluting a solution of sodium hydroxide (lye) could be technically described as acidifying it, but it will
never become acidic! Nor should one give the wrong impression by describing conditions at, for
instance, Vostok in Antarctic as “warming” when the temperature rises from -
gets warm there, just less cold.
Figure 2 shows a set of calculated values of pH for most of the world's ocean surfaces from Reference
1. As is typical of such charts, the distinction between actual measurements and imaginative simulation
is blurred. We assume, for our purposes here, that this comes close to reality, at one point in time.
Note that the lowest pH shown is about 7.9 and the highest about 8.2. There is no “acid” off the
Northern California coast and even where the pH of the ocean surface is lowest, it is still solidly basic.
In areas where large quantities of fresh water pour into the oceans (such as at the mouth of the Amazon
river), it may be possible to observe water that is slightly acidic, because fresh water is generally a little
acidic (pH less than 7). Figure 1 suggests that river water has a pH of about 6.
There are indications that corals in some areas suffer from the effects of man – but for much more
mundane reasons like mechanical damage, overfishing, water pollution, or sedimentation. (Reference
2).
Humans release about 23 million metric tons of carbon into the atmosphere every day as carbon
dioxide. Measurements of atmospheric carbon dioxide show that it is only increasing by about 11
million tons per day. If we assume that all of this increase is human caused (in line with Gore), then
12 million tons must be going elsewhere. There are two general possibilities for “elsewhere:” the
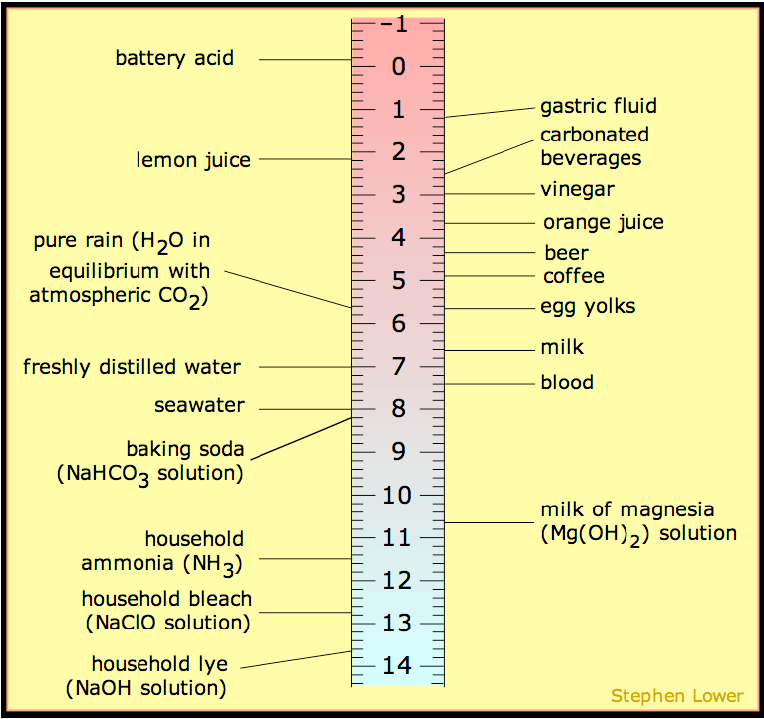
Figure 1. The pH scale showing values for typical aqueous solutions. (from Wikipedia)
biosphere and the oceans. From seasonal variations in atmospheric carbon dioxide, it is clear that the
biosphere is capable of absorbing all of this. But for our purposes here, let us assume that roughly half
disappears into the biosphere and half into the oceans. Some shows up as increased crop production,
for instance, better yields per acre for corn. That leaves about 6 million tons going into the oceans
where it likely ends up eventually sequestered in the deep ocean. Six million tons per day my seem
like a lot, but is tiny compared to the amounts naturally in play. We humans exhale about 2 million
tons of carbon per day just by breathing! If we inflate these numbers by a little more than a factor of
three by counting the atmospheric oxygen that goes into making the CO2, as Gore does, we still get
less than he claims is going into the oceans. In other words, Gore's numbers are exaggerated.
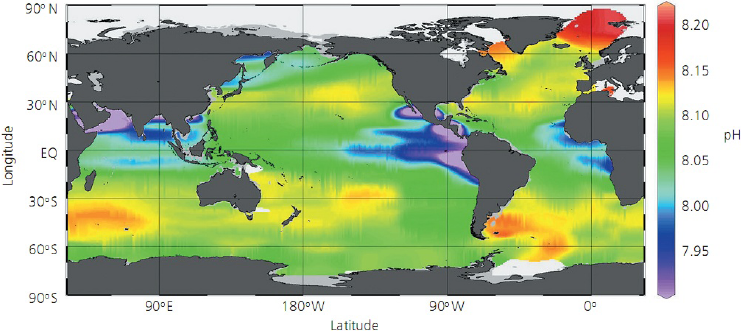
Figure 2. Calculated ocean pH (presumably based on some measurements). (Reference 1)
THE OCEAN ACIDIFICATION HYPOTHESIS according to the Royal Society
The British Royal Society presents the same hypothesis as Gore but with much more sophistication that
makes it seem more plausible:
“The oceans are absorbing carbon dioxide (CO2) from the atmosphere and this is causing chemical
changes by making them more acidic (that is, decreasing the pH of the oceans). In the past 200 years
the oceans have absorbed approximately half of the CO2 produced by fossil fuel burning and cement
production. Calculations based on measurements of the surface oceans and our knowledge of ocean
chemistry indicate that this uptake of CO2 has led to a reduction of the pH of surface seawater of 0.1
units, equivalent to a 30% increase in the concentration of hydrogen ions.
If global emissions of CO2 from human activities continue to rise on current trends then the average
pH of the oceans could fall by 0.5 units (equivalent to a three fold increase in the concentration of
hydrogen ions) by the year 2100. This pH is probably lower than has been experienced for hundreds of
millennia and, critically, this rate of change is probably one hundred times greater than at any time
over this period. The scale of the changes may vary regionally, which will affect the magnitude of the
biological effects.
Ocean acidification is essentially irreversible during our lifetimes. It will take tens of thousands of
years for ocean chemistry to return to a condition similar to that occurring at pre-
(about 200 years ago). Our ability to reduce ocean acidification through artificial methods such as the
addition of chemicals is unproven. These techniques will at best be effective only at a very local scale,
and could also cause damage to the marine environment. Reducing CO2 emissions to the atmosphere
appears to be the only practical way to minimise the risk of large-
Oceans.
All the evidence collected and modelled to date indicates that acidification of the oceans, and the
changes in ocean chemistry that accompany it, are being caused by emissions of CO2
into the atmosphere from human activities. The magnitude of ocean acidification can be predicted with
a high level of confidence. The impacts of ocean acidification on marine organisms and their
ecosystems are much less certain but it is likely that, because of their particular physiological
attributes, some organisms will be more affected than others......
Research into the impacts of high concentrations of CO2 in the oceans is in its infancy and needs to
be developed rapidly. We recommend that a major, internationally coordinated effort be launched to
include global monitoring, experimental, mesocosm and field studies. Models that include the effects
of pH at the scale of the organism and the ecosystem are also necessary. The impacts of ocean
acidification are additional to, and may exacerbate, the effects of climate change. For this reason,
the necessary funding should be additional and must not be diverted from research into climate
change.”
The last paragraph in this quotation was set in bold type by the Royal Society and needs to be read with
the impact they intended. It is both an admission that very little is really known about the effects of
atmospheric CO2 on the oceans and a plea that governments should set aside huge new sums of money
to fund research in this area. They want the funding to be in addition to the vast funding of Global
Warming so as not to be placed in competition with their friends on the Global Warming side. The last
thing they want is for Global Warming scientists to argue against their proposed ocean efforts to protect
their own research money.
Since the Royal Society paper is a proposal for huge sums of money to conduct new research, since the
basic tactic of presenting an apocalyptic view is completely self-
distinguish between what is known based on hard evidence and what is speculative theory, it is hard to
take this paper too seriously. Yet this paper is also a semi-
one in today's advocacy science. It is one of the primary sources that is quoted today. So let us look at
what they say and more importantly what they leave out or ignore that someone not seeking money
might argue.
We all agree that atmospheric carbon dioxide combines with water to form a mild acid, carbonic acid
H2CO3. This makes everything from rainwater to beer slightly acidic. But the hydration equilibrium
constant is quite small, so most of the CO2 in water remains as CO2. Carbonic acid is important for
everything from cave formation in limestone to the transport of CO2 and the buffering of pH in
mammalian blood. But H2CO3 is very unstable and readily reverts to CO2 and water. Soft drinks,
beer, and bubbly wine can be made with H2CO3 but will easily give up their carbonation if shaken or
Warmed.
According to Henry's Law the concentration of CO2 in the air above an aqueous solution of CO2
should maintain a proportionality. In other words, a doubling of atmospheric CO2 should result in a
doubling of CO2 in the aqueous solution at a constant temperature in the absence of buffering. But if
the temperature of the aqueous solution goes up, the solution can hold less gas and
will out-
its stored CO2 to the air above. This is the likely explanation for the observed lag between atmospheric
temperature and atmospheric CO2 concentration in the ice core data that shows climate history going
back almost a million years. The air warms followed by the ocean warming and giving off CO2
centuries later.
With CO2 and H2CO3 in sea water, we get bicarbonate ion production as H2CO3 splits into H+ and
HCO3-
Ca++ and CO3-
process. The carbon dioxide entering the water causes a slight decrease in pH followed by a shift back
to higher pH by the dissolution of calcium carbonate, and the uptake of hydrogen ions to form
bicarbonate ions. The oceans have vast stores of calcium carbonate available for this buffering.
Hence the ocean pH has to remain very close to 8.0. There is no net acidification even though this
scare has been given that name.
It is remarkable that alarmist scientists continue to use terminology in headlines and summaries that
they argue deeper in a paper cannot represent reality.
The concern that they raise is the dissolution of calcium carbonate in the buffering process will harm
the ability of sea creatures such as corals to grow or to maintain shells. They admit that the oceans
have an abundance of calcium carbonate for the buffering process but assert that ocean mixing is
insufficient to make the great stores in sediments available in timely fashion. Hence they argue that sea
creatures will suffer as they become the buffering material close to the ocean surface.
To make matters more complicated, calcium carbonate occurs in two crystalline forms in sea creatures:
calcite and aragonite. Aragonite is more soluble than calcite; hence sea creatures using this form of
calcium carbonate could be more easily harmed in the assumed scenario. Alarmists produce beautiful
computer graphics showing simulated ocean conditions for aragonite from before the Industrial
Revolution (1765) through the future (2100). See Figure 3. Such simulations are likely the source of
claims that ocean conditions and pH have changed markedly since the 1700s. There have certainly
been no measurements except in very recent times. The pH scale was not even invented until the early
1900s.
Even today there is no reliable, global monitoring of ocean pH, although there have been some global
measurements at one point in time as shown in Figure 2. Hence, there is no reliable way to tell whether
pH has changed significantly due to any cause, let alone to man.
If measurements had been made in prior centuries, they would likely have shown different ocean
conditions, much as we know that climate conditions have changed observably over time. But it is
ludicrous to attribute these to anthropogenic carbon dioxide in the 1700s, 1800s, or early 1900s because
the large increase in man-
study of varying conditions on earth MUST first eliminate natural causes before attributing variations
to man. Failure to do that renders the conclusions invalid.
Anthropogenic carbon dioxide is a minor player in the overall carbon cycle on the earth. Each year
man releases about 8.5 Gt of carbon into the atmosphere from the burning of coal, oil, and natural gas
as well as the production of cement and breathing. Humans exhale about 0.6 Gt of carbon
as CO2 that originated in plants. (1Gt = 10exp15 grams = 10exp12 kg = one billion metric tons).
The atmosphere as a whole contains about 780 Gt of carbon; the surface of the oceans contain about
1,000 Gt of carbon; vegetation and soils contain 2,000 Gt of carbon; and the intermediate and deep
oceans contain 38,000 Gt of carbon. Each year, the ocean surface and atmosphere exchange an
estimated 90 Gt of carbon; vegetation and the atmosphere about 100 Gt of carbon; marine biota and
the ocean surface about 50 Gt of carbon; and the ocean surface and the intermediate and deep oceans
about 40 Gt of carbon. All of these numbers are greatly larger than the human contributions,
indicating that man's effects are small but not completely insignificant. (Reference 3)
Since these natural reservoirs for carbon are highly coupled and constantly interacting in complex ways
with far greater exchanges of carbon than man's contribution to the atmosphere, it is not obvious where
the small human contribution goes. Measurements of atmospheric carbon dioxide show an increase of
only 4 Gt/year of carbon. Hence the remaining human contribution must be absorbed elsewhere. Some
(but certainly not all, as the Royal Society maintains) can be going into the oceans. Much of it is
probably going into the biosphere (on land and sea). At 388 ppm, the earth's atmosphere has a very
low concentration of carbon dioxide that inhibits plant growth. If the concentration falls below 200
ppm, plants cannot grow and will die. They prefer about 1,000 ppm. Recent increases have been
enormously beneficial.
From radioisotope studies, we know that an average molecule of carbon dioxide spends about 5 years
in the atmosphere. It is frequently estimated that half those molecules leaving the atmosphere go into
the ocean surface and half into land vegetation. But this has to vary with ocean surface temperature
and growing conditions on land. A warming ocean will naturally out-
core data) but should not out-
the “excess” man-
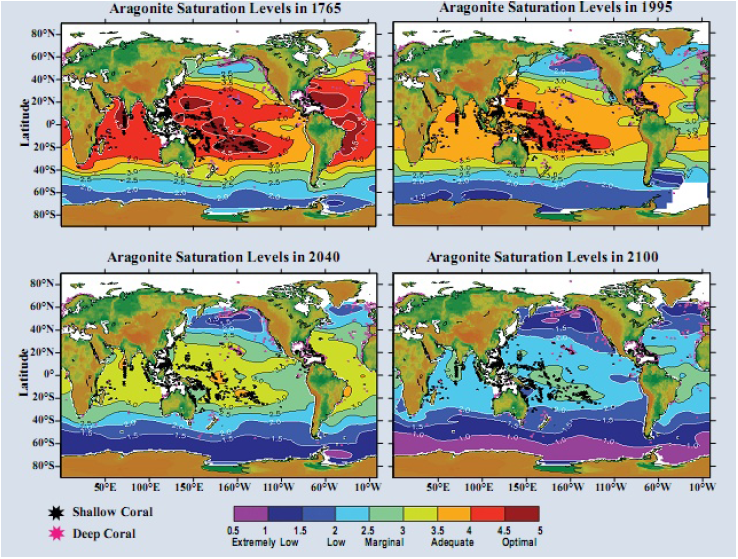
Figure 3 Simulated aragonite saturation states of the surface ocean: past, present, and future.
(Feely et al 2006 and Reference 4)
and less into solution in the oceans. Under cooling conditions, the opposite should be true.
The oceans are anything but uniform in temperature and have complex circulation patterns that make
the carbon sequestration picture much more complex. But the fact remains that the human part of the
carbon cycle is very small and can never become huge because most of the carbon on this planet (that
was probably found originally in a primordial atmosphere as CO2) is now tied up in
non-
rocks such as limestone or marble not as coal, oil, or natural gas.
Another perspective for judging the survivability of calcium carbonate creatures like coral under ocean
changes that may occur in the future (due to natural or man-
geological record. Calcite corals are known to have evolved during the Cambrian era, 550 million years
ago. At that time there was twenty times as much CO2 in the atmosphere as there is today. The more
delicate aragonite corals evolved 175 million years ago, in the Triassic era, when there was also about
twenty times as much CO2 as there is today. Corals are obviously capable of adapting to huge changes
in atmospheric CO2, whatever the resulting changes in sea water alkalinity. They have also survived
the large variations in sea level that come with each transition between an ice age and interglacial
period. When sea level drops dramatically at the onset of the next ice age, many corals will be left high
and dry to die. But those remaining will just set up housekeeping where the new conditions are most
appropriate for them. Every creature and plant on earth will do the same thing or perish.
Corals for all their delicate appearance have been extremely resilient over the earth's history, even
surviving the geologic event that killed off the dinosaurs. The far more delicate creature, that will have
much more trouble surviving natural climate variations certain to occur, is man. Of course, he has a
brain but often does not use it.
REFERENCES:
1) “Ocean acidification due to increasing carbon dioxide,” The Royal Society (policy document),
ISBN 0 85403 617 2, June 2005.
2) “Humans Are Destroying Earth's Coral Reefs …,”CO2 Science, Volume 12, Number 41: 14
October 2009.
3) “Environmental Effects of Increased Atmospheric Carbon Dioxide,”Arthur B. Robinson, Noah
E. Robinson, and Willie Soon, Oregon Institute of Science and Medicine, 2251 Dick George
Road, Cave Junction, Oregon 97523.
4) “Impacts of ocean acidification on coral reefs and other marine calcifiers,” Report of a
workshop sponsored by NSF, NOAA, USGS,” JA Kleypas, et. al., June 2006.
This paper in PDF form
| Warming Stopped |
| NOAA Data |
| NorthWest |
| Oregon |
| Washington |
| Extreme Weather |
| Past Was Warmer |
| NASA: 30's Hotter |
| PastBeliefs |
| HistoryOFAlarmism |
| Central England |
| Temperature History |
| MultiProxy |
| treemometers |
| Northwest Passage |
| Acidification-Ball |
| Acidification-Fulks |
| Acidification-Idso |
| Selected Emails |
| CRU Emails - html formatted |
| CRU Emails Simple Format |
| CRU Emails UnFormatted |
| DCPS paper |
| CRU_Files_Notice |
| False Deadlines |
| Hockey Stick Links |